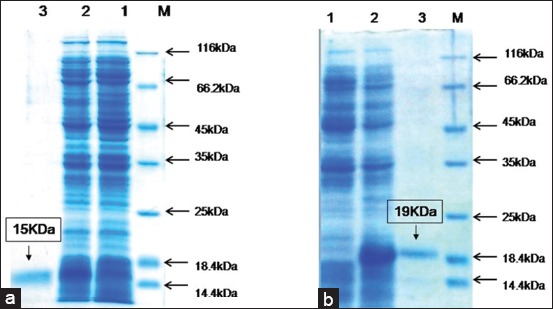
Background and ObjectivesMost Interleukin (IL) -4 and IL-10 activates immune cells and induce a humoral immune response. However, horses recombinant version of IL-4 and IL-10 has not been studied to understand their immunomodulating activities. This study aims to produce horses mature recombinant IL-4 and IL-10 in Escherichia coli. Immune-modulating activity of recombinant equine IL-4 and…